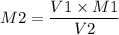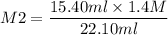If I have 15.40mL of 1.4M HBr(aq) and add 22.10mL KOH what is the molarity of
KOH being used?...

Chemistry, 07.04.2020 21:31, noathequeen
If I have 15.40mL of 1.4M HBr(aq) and add 22.10mL KOH what is the molarity of
KOH being used?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 21:40, k3rbycalilung
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2


Chemistry, 22.06.2019 23:30, Xavier8247
Rank the following four acids in order of increasing bronsted acidity : h2f+ , ch3oh, (ch3)2oh+ , ch3sh2+
Answers: 3
Do you know the correct answer?
Questions in other subjects:


Mathematics, 12.10.2020 03:01


Mathematics, 12.10.2020 03:01



Mathematics, 12.10.2020 03:01


English, 12.10.2020 03:01