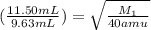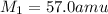If 11.50 mL of an unknown gas effuses through a small hole in a heated vessel in the same time it takes 9.63 mL of argon to effuse through the hole under the same conditions, what is the molecular weight of the unknown gas. (Use 3 sig figs and amu for the units of the answer.)

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, tifftifftiff5069
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Do you know the correct answer?
If 11.50 mL of an unknown gas effuses through a small hole in a heated vessel in the same time it ta...
Questions in other subjects:


English, 17.10.2020 14:01

Mathematics, 17.10.2020 14:01

Chemistry, 17.10.2020 14:01

Physics, 17.10.2020 14:01

Mathematics, 17.10.2020 14:01

Chemistry, 17.10.2020 14:01

English, 17.10.2020 14:01

Mathematics, 17.10.2020 14:01

Mathematics, 17.10.2020 14:01


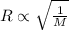
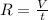
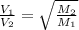
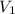 = rate of effusion of unknown gas = 11.50 mL
= rate of effusion of unknown gas = 11.50 mL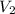 = rate of effusion of argon gas = 9.63 mL
= rate of effusion of argon gas = 9.63 mL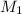 = molar mass of unknown gas
= molar mass of unknown gas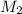 = molar mass of argon gas = 40 amu
= molar mass of argon gas = 40 amu