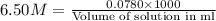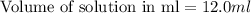Chemistry, 07.04.2020 15:52, boweytom6217
Zinc reacts with hydrochloric acid according to the reaction equation Zn ( s ) + 2 HCl ( aq ) ⟶ ZnCl 2 ( aq ) + H 2 ( g ) How many milliliters of 6.50 M HCl ( aq ) are required to react with 2.55 g Zn ( s ) ?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 01:30, AptAlbatross
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3


Chemistry, 23.06.2019 09:00, AdoNice
A2-kg bowling ball is 1 meter off the ground on a post when it falls. just before it reaches the ground, its traveling 4.4 m/s. assuming that there is no air resistant, which statement is true a. the initial potential energy is less then the final kinetic energy b. the mechanical energy is not conserved c. the mechanical energy is conserved d. the initial potential energy is greater than the final kinetic energy
Answers: 3

Chemistry, 23.06.2019 13:00, 4804344130
Aecosystem is if it can continue to function over long periods of time
Answers: 1
Do you know the correct answer?
Zinc reacts with hydrochloric acid according to the reaction equation Zn ( s ) + 2 HCl ( aq ) ⟶ ZnCl...
Questions in other subjects:

Computers and Technology, 02.09.2021 14:00



Mathematics, 02.09.2021 14:00




English, 02.09.2021 14:00

Mathematics, 02.09.2021 14:00


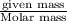
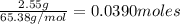
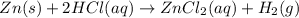
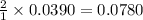 moles of HCl
moles of HCl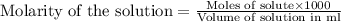 .....(1)
.....(1)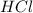 solution = 6.50 M
solution = 6.50 M