
Chemistry, 06.04.2020 01:02, winterblackburn78
According to the following equation, Calculate the percentage yield if 550.0 g of toluene (C7H8 )added to an excess of nitric acid (HNO3) provides 305 g of the p-nitrotoluene (C7H7NO2 ) product in a lab experiment.
___C7H8 + ___HNO3 ___C7H7NO2 + ___H2O

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Kaylinne1181
This chart represents the melting point of several substance. what besy explains the high melting point of the salt?
Answers: 2


Chemistry, 22.06.2019 13:30, princessroseee769
Ants live on acacia trees in south america. the ants feed on sugars secreted by the trees. the trees provide room for the ants to live. the ants sting any other insect or animal that comes to eat the trees. what type of relationship is this?
Answers: 1

Chemistry, 22.06.2019 16:00, sassy11111515
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1
Do you know the correct answer?
According to the following equation, Calculate the percentage yield if 550.0 g of toluene (C7H8 )add...
Questions in other subjects:











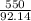
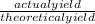 x 100
x 100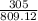 x 100
x 100




