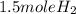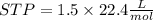Chemistry, 05.04.2020 05:17, screamqueen
2Li + H2SO4=Li2SO4 + H2 How many liters of hydrogen gas, H2 at STP can be produced from 3.0 moles of Li? The molar volume of a gas at STP is 22.4L/mol.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:50, awesomegamergurl13
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

Chemistry, 22.06.2019 19:30, gracieisweird12
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 21:30, Turtlelover05
How can the periodic table be used to predict the behavior of elements?
Answers: 1
Do you know the correct answer?
2Li + H2SO4=Li2SO4 + H2 How many liters of hydrogen gas, H2 at STP can be produced from 3.0 moles of...
Questions in other subjects:




Social Studies, 10.11.2019 00:31

English, 10.11.2019 00:31

English, 10.11.2019 00:31


Mathematics, 10.11.2019 00:31

Mathematics, 10.11.2019 00:31


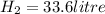
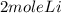 ⇔
⇔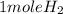
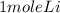 ⇔
⇔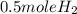
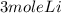 ⇔
⇔