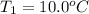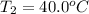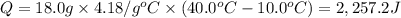Chemistry, 05.04.2020 03:16, meganwintergirl
Calculate the energy needed to raise the temperature of 18.0g of water from 10.0C to 40.0C. The specific heat of water is 4.18 J/gC. *

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, kayleg907436
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1


Chemistry, 22.06.2019 16:30, montanolumpuy
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 23.06.2019 05:00, cpcoolestkid4
C=59(f−32)the equation above shows how temperature f, measured in degrees fahrenheit, relates to a temperature c, measured in degrees celsius. based on the equation, which of the following must be true? a temperature increase of 1 degree fahrenheit is equivalent to a temperature increase of 59 degree celsius. a temperature increase of 1 degree celsius is equivalent to a temperature increase of 1.8 degrees fahrenheit. a temperature increase of 59 degree fahrenheit is equivalent to a temperature increase of 1 degree celsius. a) i onlyb) ii onlyc) iii onlyd) i and ii only
Answers: 1
Do you know the correct answer?
Calculate the energy needed to raise the temperature of 18.0g of water from 10.0C to 40.0C. The spec...
Questions in other subjects:

Mathematics, 01.07.2019 02:30

Physics, 01.07.2019 02:30




Mathematics, 01.07.2019 02:30

Mathematics, 01.07.2019 02:30


SAT, 01.07.2019 02:30

History, 01.07.2019 02:30


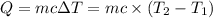
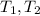 : Initial and final temperature of the substance
: Initial and final temperature of the substance