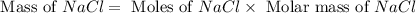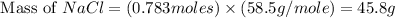2Na+Cl 2 →2NaCl2, start text, N, a, end text, plus, start text, C, l, end text, start subscript, 2, end subscript, right arrow, 2, start text, N, a, C, l, end text How many grams of \text{NaCl}NaClstart text, N, a, C, l, end text will be produced from 18.0 \text{ g}18.0 g18, point, 0, start text, space, g, end text of \text{Na}Nastart text, N, a, end text and 23.0 \text{ g}23.0 g23, point, 0, start text, space, g, end text of \text{Cl}_2Cl 2 start text, C, l, end text, start subscript, 2, end subscript?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:40, janelisse199820
Non renewable resources like petroleum eventually
Answers: 2

Chemistry, 22.06.2019 09:00, dante766
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1
Do you know the correct answer?
2Na+Cl 2 →2NaCl2, start text, N, a, end text, plus, start text, C, l, end text, start subscript, 2,...
Questions in other subjects:

English, 20.11.2020 06:20


Mathematics, 20.11.2020 06:20


Mathematics, 20.11.2020 06:20

Mathematics, 20.11.2020 06:20

Social Studies, 20.11.2020 06:20

Arts, 20.11.2020 06:20

Mathematics, 20.11.2020 06:20

Mathematics, 20.11.2020 06:20


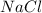 produced is, 45.8 grams.
produced is, 45.8 grams.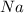 = 18.0 g
= 18.0 g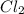 = 23.0 g
= 23.0 g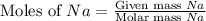
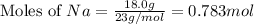
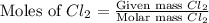
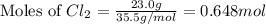
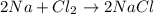
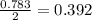 moles of
moles of