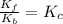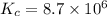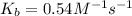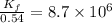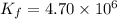Chemistry, 04.04.2020 14:31, eburnhisel2023
At 700 K the equilibrium constant KC for the reaction between NO(g) and O2(g) forming NO2(g) is 8.7 × 106. The rate constant for the reverse reaction at this temperature is 0.54 M–1s–1. What is the value of the rate constant for the forward reaction at 700 K?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:20, lindseysmith9522
Neils bohr believed that electrons orbited the nucleus in different energy levels, based on strong support from
Answers: 1

Chemistry, 23.06.2019 00:20, HernanJe6
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 00:50, alainacorkell6472
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1

Chemistry, 23.06.2019 01:00, dawnparker71
Which substance—wood or silver—is the better thermal conductor? a thermal conductor is a material that requires very little heat energy to change its temperature. explain your answer.
Answers: 3
Do you know the correct answer?
At 700 K the equilibrium constant KC for the reaction between NO(g) and O2(g) forming NO2(g) is 8.7...
Questions in other subjects:










Mathematics, 15.07.2021 19:00


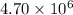
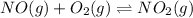
![K_c=\frac{[NO_2]}{[NO][O_2]}](/tpl/images/0582/5120/67e8f.png)
![R_f=K_f[NO][O_2]](/tpl/images/0582/5120/9cd84.png)
![R_b=K_b[NO_2]](/tpl/images/0582/5120/d91b1.png)
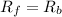
![K_f[NO][O_2]=K_b[NO_2]](/tpl/images/0582/5120/7432e.png)
![\frac{K_f}{K_b}=\frac{[NO_2]}{[NO][O_2]}](/tpl/images/0582/5120/419f2.png)