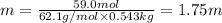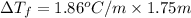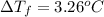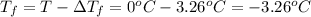Chemistry, 04.04.2020 10:57, ehgdhjahag
What is the freezing point of a solution of ethylene glycol, a nonelectrolyte, that contains 59.0 g of (CH2OH)2 dissolved in 543 g of water? Use molar masses with at least as many significant figures as the data given.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:30, samantha9430
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

Chemistry, 22.06.2019 21:20, 50057543
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

Chemistry, 23.06.2019 00:00, scottykinkade7860
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1

Chemistry, 23.06.2019 00:00, baseball1525
Which item is most likely part of the safety contract
Answers: 1
Do you know the correct answer?
What is the freezing point of a solution of ethylene glycol, a nonelectrolyte, that contains 59.0 g...
Questions in other subjects:


Mathematics, 21.06.2019 19:20


Mathematics, 21.06.2019 19:20

Geography, 21.06.2019 19:20

Mathematics, 21.06.2019 19:20





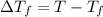
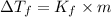
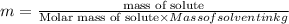
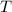 = Freezing point of solvent
= Freezing point of solvent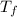 = Freezing point of solution
= Freezing point of solution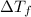 =depression in freezing point
=depression in freezing point 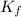 = freezing point constant
= freezing point constant