
Chemistry, 04.04.2020 11:05, ellaemtagedeane
When iron(III) oxide reacts with aluminum, aluminum oxide and iron are produced. The balanced equation for this reaction is:
Fe2O3 (s) + 2Al (s) > Al2O3 (s) + 2Fe (s)
If 6 moles of aluminum react:
i. The reaction consumes moles of iron(III) oxide.
ii. The reaction produces moles of aluminum oxide and moles of iron.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, portedon8644
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3

Chemistry, 22.06.2019 06:40, alyons60
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 11:40, jerrysandoval22
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Do you know the correct answer?
When iron(III) oxide reacts with aluminum, aluminum oxide and iron are produced. The balanced equati...
Questions in other subjects:

Mathematics, 17.06.2021 20:30

Biology, 17.06.2021 20:30

Mathematics, 17.06.2021 20:30




Chemistry, 17.06.2021 20:30



Mathematics, 17.06.2021 20:30


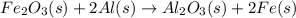
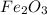
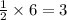 moles of
moles of 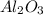
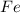
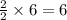 moles of
moles of 



