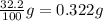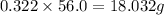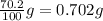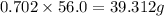Chemistry, 04.04.2020 10:59, iklassibrahim123
Consider the solubilities of a particular solute at two different temperatures. Temperature ( ∘ C ) Solubility ( g / 100 g H 2 O ) 20.0 32.2 30.0 70.2 Suppose a saturated solution of this solute was made using 56.0 g H 2 O at 20.0 °C. How much more solute can be added if the temperature is increased to 30.0 ∘ C? mass:

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:20, phanuel642
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2

Chemistry, 22.06.2019 20:30, huangjianhe135
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 23.06.2019 00:20, jessicamcummins
What type of context clue you understand the meaning of quandary?
Answers: 3
Do you know the correct answer?
Consider the solubilities of a particular solute at two different temperatures. Temperature ( ∘ C )...
Questions in other subjects:

Geography, 27.02.2021 01:00





Arts, 27.02.2021 01:00


Mathematics, 27.02.2021 01:00