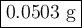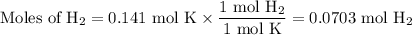Chemistry, 03.04.2020 06:24, spdesch2558
2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hydrogen gas(H₂)
●a). What is the limiting reactant?
●b.)What is the excess reactant?
●C.)How much product is produced?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:50, zaleemawhite
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 08:30, myamiller558
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 13:00, yaneiryx5476
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1
Do you know the correct answer?
2K + 2HBr → 2 KBr + H2
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hyd...
When 5.5moles of K reacts with 4.04moles of HBr, to produce Hyd...
Questions in other subjects:

English, 04.10.2021 19:10



Mathematics, 04.10.2021 19:10

English, 04.10.2021 19:10


Mathematics, 04.10.2021 19:10



History, 04.10.2021 19:10