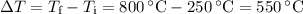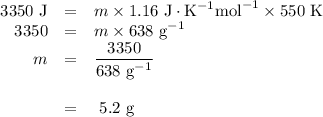Chemistry, 03.04.2020 06:38, Jazongamez1987
3350 J of heat is required to raise the temperature of a sample of AlF3 from 250C to 800C. What is the mass of the sample?
I just want to know how to work this out. Please help me.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:30, jonloya264
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 20:00, 20calzoy
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
Do you know the correct answer?
3350 J of heat is required to raise the temperature of a sample of AlF3 from 250C to 800C. What is t...
Questions in other subjects:

Mathematics, 06.06.2020 05:02


History, 06.06.2020 05:02


History, 06.06.2020 05:02



Mathematics, 06.06.2020 05:02