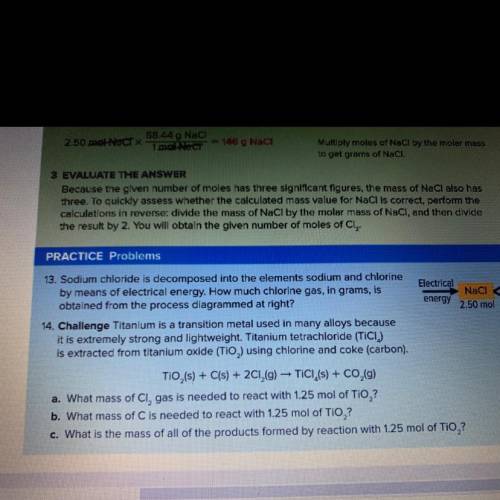
What is the mass of all the products formed by the reaction with 1.25 mol of TiO2?
PLEASE HELP...

Chemistry, 03.04.2020 06:05, chayaharroch03
What is the mass of all the products formed by the reaction with 1.25 mol of TiO2?
PLEASE HELP ASAP

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:00, joelpimentel
This flow chart shows the amount of energy that is emitted by each type of light. ultraviolet > blue light > yellow light > red light (maximum energy) (minimum energy) in an experiment, shining which type of light on a strip of metal would be least likely to produce the photoelectric effect? ultraviolet light dim blue light bright red light bright yellow light
Answers: 2

Chemistry, 22.06.2019 22:00, allieeastridge
For a family dinner, jose’s mom baked a loaf of bread. during the meal, his grandmother commented, “this bread is so dense! ” what do you think his grandmother meant by the word dense? you have some ideas regarding density already, but this experiment will enrich your understanding of the relationship between mass, volume and the density of objects. when making measurements in this experiment we will be using si units, the international system of units. the purpose of using si units is to have the ability to communicate and compare data results with other experiments without having to make conversions. measurement symbol si unit length m meter mass kg kilogram volume l liters density kg/l kilograms per liter any calculations that are performed during this experiment will ultimately be reported in scientific notation. recall that scientific notation is extremely important when we have data that is extremely small or large. scientists rely on a standard form of communication to quickly make hypotheses and judgements based on data. what is the density of block a? a0kg/l what is the density of block b? a1kg/l what is the density of block c? a2kg/l what is the density of block d? a3kg/l what is the density of block e? a4kg/l which block is the densest? a5
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Mathematics, 25.07.2019 10:00


History, 25.07.2019 10:00


Mathematics, 25.07.2019 10:00




Advanced Placement (AP), 25.07.2019 10:00







