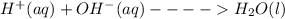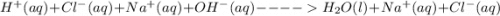Chemistry, 03.04.2020 02:44, josephvaldez518
HCl(aq)+NaOH(aq)→NaCl(aq)+H2O(l)
A student was given the task of titrating a 20.mL sample of 0.10MHCl(aq) with 0.10MNaOH(aq) . The HCl(aq) was placed in an Erlenmeyer flask. An equation for the reaction that occurs during the titration is given above.
(b) The equation above is not written in net ionic form. Write the correct net ionic equation for the reaction.

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 08:00, flakko1899
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 10:00, berniceallonce22
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1
Do you know the correct answer?
HCl(aq)+NaOH(aq)→NaCl(aq)+H2O(l)
A student was given the task of titrating a 20.mL samp...
A student was given the task of titrating a 20.mL samp...
Questions in other subjects:


Mathematics, 29.01.2021 22:50

English, 29.01.2021 22:50

Mathematics, 29.01.2021 22:50


Mathematics, 29.01.2021 22:50




History, 29.01.2021 22:50