
Chemistry, 02.04.2020 22:14, bobbyhill24
A 7.11g sample of potassium chlorate was decomposed according to the following equation: 2KCLO3 -->2KCL + 3O2 How many moles of oxygen are formed?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, hannah5143
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 17:40, aaliyahthomas37
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 20:00, 20calzoy
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
Do you know the correct answer?
A 7.11g sample of potassium chlorate was decomposed according to the following equation: 2KCLO3 --&g...
Questions in other subjects:

Mathematics, 28.04.2021 04:50



Mathematics, 28.04.2021 04:50

Mathematics, 28.04.2021 04:50

Mathematics, 28.04.2021 04:50


Mathematics, 28.04.2021 04:50


History, 28.04.2021 04:50


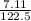
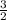 × 0.0580 moles
× 0.0580 moles



