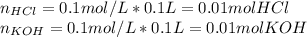Chemistry, 01.04.2020 16:47, maxi12312345
As 100 milliliters of 0.10 molar KOH is added to 100 milliliters of 0.10 molar HCL at 298 K, the pH of the res?A. decrease to 3B. decrease to 4C. increase to 7D. increase to 13

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:00, adrian128383
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 22:30, creepycrepes
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1
Do you know the correct answer?
As 100 milliliters of 0.10 molar KOH is added to 100 milliliters of 0.10 molar HCL at 298 K, the pH...
Questions in other subjects:

Chemistry, 19.04.2020 20:59

Spanish, 19.04.2020 20:59




Mathematics, 19.04.2020 20:59

Mathematics, 19.04.2020 20:59

Mathematics, 19.04.2020 20:59