
Chemistry, 01.04.2020 13:36, genyjoannerubiera
Calculate the volume occupied by 272g
of methane at a pressure of 250 kPa and a
temperature of 54°C.
(R = 8.31JK-1 mol-1; M, methane = 16.0)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, ayaanwaseem
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2


Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 23:00, lufung8627
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
Do you know the correct answer?
Calculate the volume occupied by 272g
of methane at a pressure of 250 kPa and a
temperatur...
of methane at a pressure of 250 kPa and a
temperatur...
Questions in other subjects:

English, 22.08.2021 17:30

History, 22.08.2021 17:30

Mathematics, 22.08.2021 17:30

History, 22.08.2021 17:30

Mathematics, 22.08.2021 17:30

History, 22.08.2021 17:30

English, 22.08.2021 17:30

Computers and Technology, 22.08.2021 17:30


Mathematics, 22.08.2021 17:30


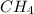 ) = 272 grams
) = 272 grams 



