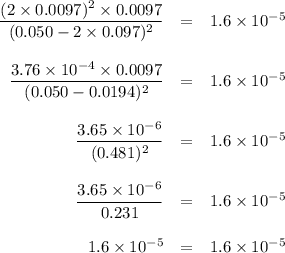Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, mimireds5419
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3


Chemistry, 23.06.2019 00:30, mathwiznot45
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1

Chemistry, 23.06.2019 06:20, cowboo5000pcl655
An object of mass 10.0 kg and volume 1000 ml and density 10 g/ml sinks in water who’s density is 1.0 g/ml. what is the mass of the water which has been displaced in kilograms
Answers: 1
Do you know the correct answer?
2NOCI (g)
→ 2NO(g) + Cl2 (g)
Problem:
Initially
the above reaction contains<...
→ 2NO(g) + Cl2 (g)
Problem:
Initially
the above reaction contains<...
Questions in other subjects:







Social Studies, 27.07.2019 18:30




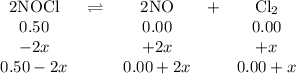
![K_{\text{c}} = \dfrac{\text{[NO]$^{2}$[Cl$_{2}$]}}{\text{[NOCl]}^{2}} = \dfrac{(2x)^{2}(x)}{(0.50 - 2x)^{2}} = 1.6 \times 10^{-5}\\\\4x^{3} = 1.6 \times 10^{-5}(0.50 - 2x)^{2}\\x^{3} = 4.0 \times 10^{-6}(0.50 - 2x)^{2}](/tpl/images/0575/0584/940ed.png)
![x = \sqrt [3] {4.0 \times 10^{-6}(0.50)^{2}} = 0.010](/tpl/images/0575/0584/0f7f4.png)
![x = \sqrt [3] {4.0 \times 10^{-6}(0.50 - 2\times 0.010)^{2}} = 0.0097](/tpl/images/0575/0584/39746.png)
![x = \sqrt [3] {4.0 \times 10^{-6}(0.50 - 2\times 0.0097)^{2}} = 0.0097](/tpl/images/0575/0584/5e7db.png)