
Chemistry, 01.04.2020 01:55, olivasm5626
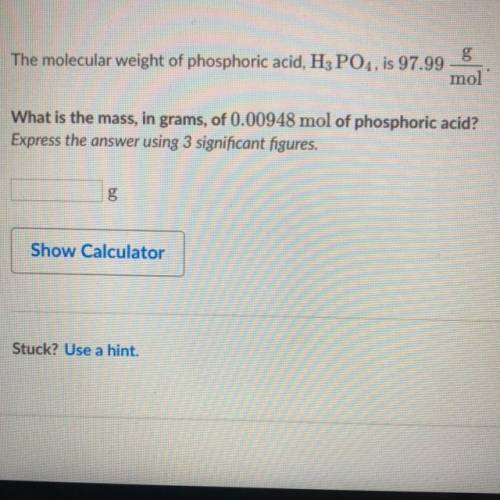
The molecular weight of phosphoric acid, H 3 PO 4 , is 97.99 * g/(mol) What is the mass, in grams, of 0.00948 mol of phosphoric acid?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:00, slugmilk1090
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1

Chemistry, 22.06.2019 17:30, ander67061
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 22.06.2019 20:20, catchonyet
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1
Do you know the correct answer?
The molecular weight of phosphoric acid, H 3 PO 4 , is 97.99 * g/(mol) What is the mass, in grams, o...
Questions in other subjects:

English, 04.03.2021 19:30



Mathematics, 04.03.2021 19:30



Mathematics, 04.03.2021 19:30


Mathematics, 04.03.2021 19:30

Biology, 04.03.2021 19:30







