
Chemistry, 01.04.2020 01:04, ericadawn2852
William measures a test tube and finds that the mass of the test tube is 5g. He places the lone reactant in the test tube and finds the mass to be 12.5g. He carries out a reaction by heating the test tube. At the end of the reaction, he measures the mass of the test tube containing the lone product and finds it to have a mass of 9.3g. What is the percent yield for this experiment if he expected to produce 5g of product?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, ashlpiriz123
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1

Chemistry, 22.06.2019 12:00, BreBreDoeCCx
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal. b. he is determining chemical properties that are sufficient to identify the metal. c. he is determining physical properties that are insufficient to identify the metal. d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 22.06.2019 16:00, sassy11111515
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1
Do you know the correct answer?
William measures a test tube and finds that the mass of the test tube is 5g. He places the lone reac...
Questions in other subjects:

Mathematics, 08.11.2020 23:10


English, 08.11.2020 23:10

Mathematics, 08.11.2020 23:10

English, 08.11.2020 23:10

English, 08.11.2020 23:10

Mathematics, 08.11.2020 23:10

History, 08.11.2020 23:10

History, 08.11.2020 23:10

English, 08.11.2020 23:10


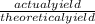 x 100
x 100 x 100
x 100



