2. If I have 45 g of Carbon, to find the number of moles of carbon I would *
divide by molar m...

Chemistry, 31.03.2020 16:16, heavendl13
2. If I have 45 g of Carbon, to find the number of moles of carbon I would *
divide by molar mass (12.01 g)
multiply by molar mass (12.01 g)
divide by Avogadro's number (6.02 x 10^23)
multiply by Avogadro's number (6.02 x 10^23)
Sushmit

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, dimondqueen511
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 13:00, monkeyrose1999
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 13:50, aesthetickait
How does the motion of particles in a gas change as the gas cools
Answers: 2
Do you know the correct answer?
Questions in other subjects:

English, 01.07.2019 13:00



Mathematics, 01.07.2019 13:00

Mathematics, 01.07.2019 13:00


Chemistry, 01.07.2019 13:00

Mathematics, 01.07.2019 13:00

Mathematics, 01.07.2019 13:00

Mathematics, 01.07.2019 13:00


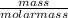
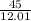 = 3.75moles
= 3.75moles 



