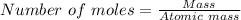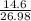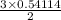Chemistry, 31.03.2020 04:37, chantianabess36
Sulfuric acid dissolves aluminum metal according to the following reaction: 2Al(s)+3H2SO4(aq)→Al2(SO4)3(aq)+3H2 (g)2Al(s)+3H2SO4(aq)→Al2(SO4)3(aq)+ 3H2(g) Suppose you wanted to dissolve an aluminum block with a mass of 14.6 gg . Part A What minimum mass of H2SO4H2SO4 would you need? Express your answer in grams.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, mastershadow2018
Agroup of students is studying convection currents. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other in an area with warm air. after 10 minutes, the balloons are released from a height of 1 meter. which of the following do the students most likely observe? a. the balloons both rise. the cold balloon is larger than the warm balloon. b. the balloons rise at the same rate. both balloons are the same size. c. the warm balloon expands and rises. the cold balloon shrinks and sinks. d. the cold balloon expands and rises. the warm balloon shrinks and sinks.
Answers: 2


Chemistry, 23.06.2019 04:40, twinchristiansp4xhd2
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1

Do you know the correct answer?
Sulfuric acid dissolves aluminum metal according to the following reaction: 2Al(s)+3H2SO4(aq)→Al2(SO...
Questions in other subjects:

Mathematics, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01

Spanish, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01

Mathematics, 18.09.2020 09:01