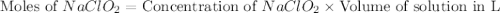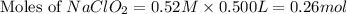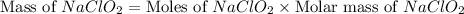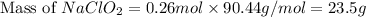Chemistry, 31.03.2020 01:01, nestergurl101
A chemistry graduate student is given 500.mL of a 0.40M chlorous acid HClO2 solution. Chlorous acid is a weak acid with =Ka×1.110−2. What mass of NaClO2 should the student dissolve in the HClO2 solution to turn it into a buffer with pH =2.11?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:30, rosieposie27
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

Do you know the correct answer?
A chemistry graduate student is given 500.mL of a 0.40M chlorous acid HClO2 solution. Chlorous acid...
Questions in other subjects:


History, 27.01.2021 02:40









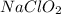 should be, 23.5 grams.
should be, 23.5 grams.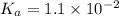
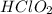 = 0.40 M
= 0.40 M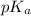 .
.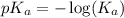
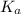 in this expression, we get:
in this expression, we get: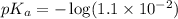
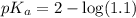
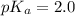
![pH=pK_a+\log \frac{[Salt]}{[Acid]}](/tpl/images/0572/2296/e961a.png)
![pH=pK_a+\log \frac{[NaClO_2]}{[HClO_2]}](/tpl/images/0572/2296/a8df0.png)
![2.11=2.0+\log (\frac{[NaClO_2]}{0.40})](/tpl/images/0572/2296/3a0b0.png)
![[NaClO_2]=0.52M](/tpl/images/0572/2296/2adfa.png)