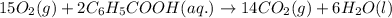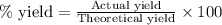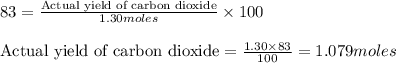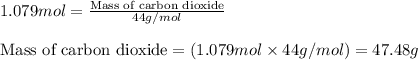Consider the following balanced equation for the following reaction:
15O2(g) + 2C6H5COOH(aq) →...

Chemistry, 31.03.2020 00:12, nicollexo21
Consider the following balanced equation for the following reaction:
15O2(g) + 2C6H5COOH(aq) → 14CO2(g) + 6H2O(l)
Determine the amount of CO2(g) formed in the reaction if the percent yield of CO2(g) is 83.0% and the theoretical yield of CO2(g) is 1.30 moles.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, minstcordell4115
Covalent network solids typically have melting points and boiling points. the chemical formula of a network solid indicates in the molecule.
Answers: 3


Do you know the correct answer?
Questions in other subjects:

Mathematics, 23.12.2020 09:00

Mathematics, 23.12.2020 09:00

English, 23.12.2020 09:00

Mathematics, 23.12.2020 09:00

Mathematics, 23.12.2020 09:00

Mathematics, 23.12.2020 09:00


Mathematics, 23.12.2020 09:00

Mathematics, 23.12.2020 09:00