
Chemistry, 30.03.2020 23:54, dontcareanyonemo
Which change to an equilibrium mixture of this reaction results in the formation of more H2S? Which change to an equilibrium mixture of this reaction results in the formation of more ? a decrease in the volume of the reaction vessel (at constant temperature) an increase in temperature an increase in the amount of NH4HS in the reaction vessel all of the above

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, cxttiemsp021
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 23.06.2019 08:00, codybrocs9624
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 2
Do you know the correct answer?
Which change to an equilibrium mixture of this reaction results in the formation of more H2S? Which...
Questions in other subjects:




Geography, 08.12.2021 20:30


Mathematics, 08.12.2021 20:30

Business, 08.12.2021 20:30



Mathematics, 08.12.2021 20:30


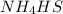 is endothermic:
is endothermic:
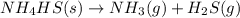
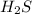 ?
?




