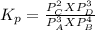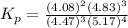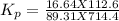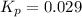At 25 ∘ C , the equilibrium partial pressures for the reaction 3 A ( g ) + 4 B ( g ) − ⇀ ↽ − 2 C ( g ) + 3 D ( g ) were found to be P A = 4.47 atm, P B = 5.17 atm, P C = 4.08 atm, and P D = 4.83 atm. What is the standard change in Gibbs free energy of this reaction at 25 ∘ C ?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, carlybeavers50
The graph above shows how the price of cell phones varies with the demand quantity. the equilibrium price for cell phones is where both supply and demand quantities equal $100, 5,000 5,000, $100
Answers: 2

Chemistry, 21.06.2019 22:20, Brooke7644
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 03:30, jabper5522
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 09:30, mimibear2932
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1
Do you know the correct answer?
At 25 ∘ C , the equilibrium partial pressures for the reaction 3 A ( g ) + 4 B ( g ) − ⇀ ↽ − 2 C ( g...
Questions in other subjects:



Mathematics, 05.01.2020 00:31

Chemistry, 05.01.2020 00:31

Mathematics, 05.01.2020 00:31

Mathematics, 05.01.2020 00:31

Social Studies, 05.01.2020 00:31



History, 05.01.2020 00:31