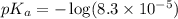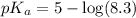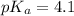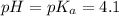The half‑equivalence point of a titration occurs half way to the equivalence point, where half of the analyte has reacted to form its conjugate, and the other half still remains unreacted. If 0.440 moles of a monoprotic weak acid ( K a = 8.3 × 10 − 5 ) is titrated with NaOH , what is the pH of the solution at the half‑equivalence point?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, Blaise2653
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1


Chemistry, 22.06.2019 20:00, jalenevoyles
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 21:30, emmalucilleblaha1995
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
Do you know the correct answer?
The half‑equivalence point of a titration occurs half way to the equivalence point, where half of th...
Questions in other subjects:


Mathematics, 05.06.2021 09:10

Social Studies, 05.06.2021 09:10

Mathematics, 05.06.2021 09:10


Mathematics, 05.06.2021 09:10

History, 05.06.2021 09:10



Mathematics, 05.06.2021 09:10


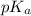 of weak acid.
of weak acid.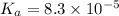
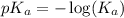
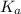 in this expression, we get:
in this expression, we get: