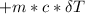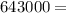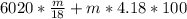Chemistry, 30.03.2020 22:31, jaccamoboy
A backpacker collects snow at 0°C, and places it in a cooking pot on a camp stove. It takes 643 kJ of heat energy to melt the snow and bring the water to boiling. Assuming no heat loss, and neglecting the specific heat capacity of the pot, calculate the mass of snow that the backpacker collected. (Data: specific heat capacity of liquid water, c = 4.18 J/g⋅K; and: H2O(s) → H2O(l) ΔH = ΔHfusion = 6.02 kJ/mol)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, girly37
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3


Do you know the correct answer?
A backpacker collects snow at 0°C, and places it in a cooking pot on a camp stove. It takes 643 kJ o...
Questions in other subjects:

Biology, 13.09.2019 19:20






Mathematics, 13.09.2019 19:20








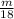 for the number of moles; so:
for the number of moles; so: