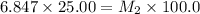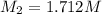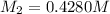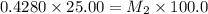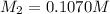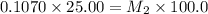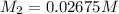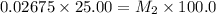Chemistry, 30.03.2020 21:08, studentms5313
Starting with a 6.847 M stock solution of HNO3, five standard solutions are prepared via serial dilution. At each stage, 25.00 mL of solution are diluted to 100.00 mL. Determine the concentration of and the number of moles of HNO3 in the final (most dilute, Md5) solution.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, jamesnaquan132
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 01:10, mistiehaas
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Do you know the correct answer?
Starting with a 6.847 M stock solution of HNO3, five standard solutions are prepared via serial dilu...
Questions in other subjects:

Mathematics, 18.03.2021 01:40





Mathematics, 18.03.2021 01:40

Chemistry, 18.03.2021 01:40




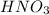 in the final solution is 0.006688 M and number of moles are 0.00006688
in the final solution is 0.006688 M and number of moles are 0.00006688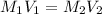
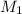 = molarity of stock solution = 6.847 M
= molarity of stock solution = 6.847 M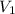 = volume of stock solution = 25.00 ml
= volume of stock solution = 25.00 ml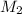 = molarity of ist dilute solution = ?
= molarity of ist dilute solution = ?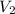 = volume of first dilute solution = 100.0 ml
= volume of first dilute solution = 100.0 ml