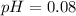Chemistry, 30.03.2020 21:10, Franklyn3220
A solution is made by adding 0.380 gg Ca(OH)2(s)Ca(OH)2(s), 50.0 mLmL of 1.45 MM HNO3HNO3, and enough water to make a final volume of 75.0 mLmL. Part A Assuming that all of the solid dissolves, what is the pH of the final solution

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:20, pilarmonsivais
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3

Chemistry, 22.06.2019 09:20, kevinhernandez582
What will most likely happen when two bromine atoms bond together?
Answers: 3
Do you know the correct answer?
A solution is made by adding 0.380 gg Ca(OH)2(s)Ca(OH)2(s), 50.0 mLmL of 1.45 MM HNO3HNO3, and enoug...
Questions in other subjects:

Arts, 04.12.2020 02:20

Engineering, 04.12.2020 02:20



English, 04.12.2020 02:20

Engineering, 04.12.2020 02:20


Mathematics, 04.12.2020 02:20


Social Studies, 04.12.2020 02:20


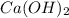 =
=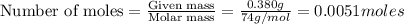
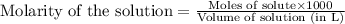
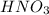 solution = 1.45 M
solution = 1.45 M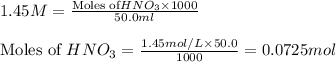
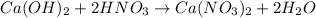
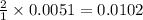 moles of
moles of 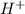 are left in 75.0 ml of solution
are left in 75.0 ml of solution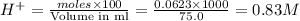
![pH=-\log [H^+]](/tpl/images/0571/2204/37e81.png)
![pH=-\log[0.83]](/tpl/images/0571/2204/f1115.png)