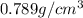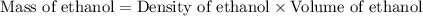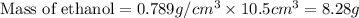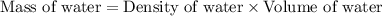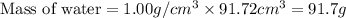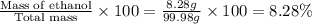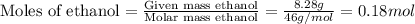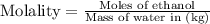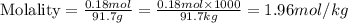Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 23:50, josie311251
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 04:40, laurabwhiddon
Equal numbers of moles of he(g), ar(g), and ne(g) are placed in a glass vessel at room temperature. if the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial pressures of the gases remaining in the vessel after some of the gas mixture has effused?
Answers: 1
Do you know the correct answer?
A bottle of wine contains 10.5% ethanol by volume. The density of ethanol (C2H5OH) is 0.789 g/cm3. C...
Questions in other subjects:





Mathematics, 25.06.2020 08:01

Mathematics, 25.06.2020 08:01

History, 25.06.2020 08:01

History, 25.06.2020 08:01


Mathematics, 25.06.2020 08:01