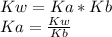Chemistry, 30.03.2020 20:27, viktoria1198zz
If you know Kb for ammonia, NH3, you can calculate the equilibrium constant, Ka, for the following reaction: NH4+ NH3 + H+ by the equation: Ka = 1 / Kb Ka = Kb / Kw Ka = Kw / Kb Ka = Kw × Kb None of these choices are correct.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, emmalybrown
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1

Chemistry, 22.06.2019 14:30, Priskittles
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1
Do you know the correct answer?
If you know Kb for ammonia, NH3, you can calculate the equilibrium constant, Ka, for the following r...
Questions in other subjects:



History, 19.03.2020 04:03


English, 19.03.2020 04:03

History, 19.03.2020 04:03