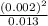Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, 5041
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1


Chemistry, 22.06.2019 05:30, sethjohnson386pbnm3x
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3
Do you know the correct answer?
0.030 moles of a weak acid, HA, was dissolved in 2.0 L of water to form a solution. At equilibrium,...
Questions in other subjects:

Spanish, 14.07.2021 16:30



English, 14.07.2021 16:30

Mathematics, 14.07.2021 16:40


Mathematics, 14.07.2021 16:40



Mathematics, 14.07.2021 16:40


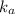 for the weak acid is
for the weak acid is 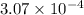 .
.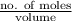
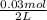
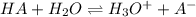
![k_{a} = \frac{[A^{-}][H_{3}O^{+}]}{[HA]}](/tpl/images/0571/0012/aea8e.png)
![\frac{x^{2}}{[HA]}](/tpl/images/0571/0012/57461.png)