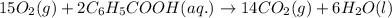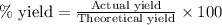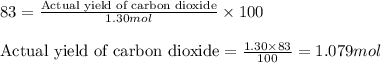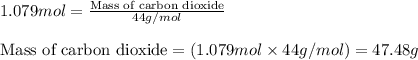Consider the following balanced equation for the following reaction:
15O2(g) + 2C6H5COOH(aq) →...

Chemistry, 30.03.2020 20:24, gibbss80stu
Consider the following balanced equation for the following reaction:
15O2(g) + 2C6H5COOH(aq) → 14CO2(g) + 6H2O(l)
Determine the amount of CO2(g) formed in the reaction if the percent yield of CO2(g) is 83.0% and the theoretical yield of CO2(g) is 1.30 moles.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, petriajack8375
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3

Chemistry, 22.06.2019 01:30, arodavoarodavo
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3

Chemistry, 22.06.2019 11:00, 21villalobosjabez
Which type of fossil does this image depict?
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Computers and Technology, 25.06.2021 16:10

Business, 25.06.2021 16:10





Biology, 25.06.2021 16:20