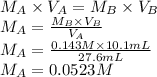Chemistry, 30.03.2020 19:38, cheaterman4121
A student is asked to standardize a solution of potassium hydroxide. He weighs out 1.08 g potassium hydrogen phthalate (KHC8H4O4, treat this as a monoprotic acid). It requires 36.8 mL of potassium hydroxide to reach the endpoint. A. What is the molarity of the potassium hydroxide solution? M This potassium hydroxide solution is then used to titrate an unknown solution of perchloric acid. B. If 10.1 mL of the potassium hydroxide solution is required to neutralize 27.6 mL of perchloric acid, what is the molarity of the perchloric acid solution? M

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, fordkenae
24 points and brainliest to anyone who can answer under 10 minutes with best ! the table below shows the role of different substances during photosynthesis. substance role during photosynthesis glucose stores chemical energy water combines with glucose to form carbon dioxide chlorophyll traps sunlight which of the following statements would correct one of the roles listed in the table? glucose combines with carbon to form water. chlorophyll reacts with light to produce carbon dioxide. water combines with carbon dioxide during photosynthesis. chlorophyll stores chemical energy needed for photosynthesis.
Answers: 1

Chemistry, 22.06.2019 04:00, breannaasmith1122
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Do you know the correct answer?
A student is asked to standardize a solution of potassium hydroxide. He weighs out 1.08 g potassium...
Questions in other subjects:

Mathematics, 14.04.2021 15:20

Mathematics, 14.04.2021 15:20

English, 14.04.2021 15:20

Mathematics, 14.04.2021 15:20


Mathematics, 14.04.2021 15:20


Biology, 14.04.2021 15:20


Mathematics, 14.04.2021 15:20