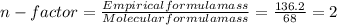A 5.769 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis and 14.92 grams of CO2 and 3.054 grams of H2O are produced. In a separate experiment, the molar mass is found to be 136.2 g/mol. Determine the empirical formula and the molecular formula of the organic compound.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:50, lejeanjamespete1
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Do you know the correct answer?
A 5.769 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis...
Questions in other subjects:



World Languages, 22.08.2021 15:10

Mathematics, 22.08.2021 15:10



Geography, 22.08.2021 15:10


English, 22.08.2021 15:10

Biology, 22.08.2021 15:10


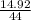 = 0.339 moles of carbonno. of moles of H₂O =
= 0.339 moles of carbonno. of moles of H₂O = 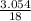 = 0.16 moles x 2 = 0.339 moles of hydrogenmass of carbon = 0.339 x 12 = 4.068 gmass of hydrogen = 0.339 x 1 = 0.339 gmass of oxygen = 5.769 - (4.068 + 0.339)
= 0.16 moles x 2 = 0.339 moles of hydrogenmass of carbon = 0.339 x 12 = 4.068 gmass of hydrogen = 0.339 x 1 = 0.339 gmass of oxygen = 5.769 - (4.068 + 0.339)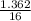 = 0.085 moles
= 0.085 moles 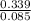 = 4relative mole ratio for hydrogen =
= 4relative mole ratio for hydrogen = 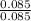 = 1 empirical formula C₄H₄Oempirical formula mass = 4 x 12 + 4 x 1 + 1 x 16 = 68molecular mass = 136.2 g / mol
= 1 empirical formula C₄H₄Oempirical formula mass = 4 x 12 + 4 x 1 + 1 x 16 = 68molecular mass = 136.2 g / mol