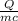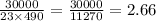Chemistry, 29.03.2020 19:47, jennainglish
30,000 J of heat are added to 23.0 kg of steel to reach a final temperature of 140
°C. What was the initial temperature of the steel? (csteel =490J/kg * °C)
1. First find AT using the formula q=mcAT
2. Rearrange to solve for initial temperature using AT=Tfinal -Tinital
I need help quickly

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, homeschool0123
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 07:40, caleb19moody
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2


Chemistry, 22.06.2019 09:00, kkmonsterhigh18
The diagram below shows a cell placed in a solution. a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution. only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it. it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2
Do you know the correct answer?
30,000 J of heat are added to 23.0 kg of steel to reach a final temperature of 140
°C. What wa...
°C. What wa...
Questions in other subjects:


Arts, 14.07.2019 18:30




Mathematics, 14.07.2019 18:30




Mathematics, 14.07.2019 18:30