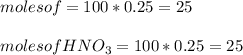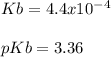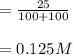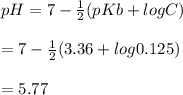A 100 mL sample of 0.25 M CH3NH2(aq) is titrated with a 100 mL of 0.25 M HNO3(aq). Select ALL main components (greater than 0.001 moles, besides H2O) that would be present in the solution after adding HNO3. Kb of CH3NH2 is 4.4 LaTeX: \times×10−4.
OH−
CH3NH2
NO3−
CH3NH3+

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, cbelew0001ouje4i
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1


Chemistry, 22.06.2019 12:30, americanbellabeauty
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

Chemistry, 23.06.2019 01:00, davelopez979
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1
Do you know the correct answer?
A 100 mL sample of 0.25 M CH3NH2(aq) is titrated with a 100 mL of 0.25 M HNO3(aq). Select ALL main c...
Questions in other subjects:

Mathematics, 28.09.2021 18:50



Chemistry, 28.09.2021 18:50






Mathematics, 28.09.2021 18:50


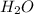 ) that would be present in the solution after adding
) that would be present in the solution after adding 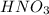 ."
." 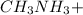 would be present in the solution after adding
would be present in the solution after adding 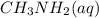 is titrated with a 100 mL of 0.25 M
is titrated with a 100 mL of 0.25 M 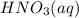 .
.