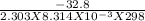Chemistry, 26.03.2020 23:00, daphnevlogs11
The Haber process for the production of ammonia is the main industrial process of producing ammonia today. Prior to developing this process, ammonia was difficult to produce on an industrial scale. The reaction for the Haber process is: N2(g) + 3 H2(g) ⇌ 2 NH3(g). Using the following values, determine the equilibrium constant for this reaction at 25oC. Substance ΔGfo(kJ/mol) N2 0 H2 0 NH3 -16.4

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:30, TaraC
Read the claim. breakfast is an important meal. it jump starts the body’s process of using calories to break down food. appetite can decrease with age, but going too long without eating causes metabolism to slow down. current research shows that incorporating legumes such as lentils and chickpeas into meals boosts metabolism for twenty-four hours. who might benefit from this claim? people who have a fast metabolism stores that sell exercise equipment people who take vitamin supplements grocery stores that sell legumes
Answers: 1

Chemistry, 23.06.2019 01:30, AptAlbatross
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3

Chemistry, 23.06.2019 03:00, rhianna18
In november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 1
Do you know the correct answer?
The Haber process for the production of ammonia is the main industrial process of producing ammonia...
Questions in other subjects:



Mathematics, 15.01.2021 02:30

Biology, 15.01.2021 02:30

Mathematics, 15.01.2021 02:30


Mathematics, 15.01.2021 02:30