
Chemistry, 26.03.2020 21:35, CHENDESHEN
A compound is found to contain 9.224 % boron and 90.74 % chlorine by mass. To answer the question, enter the elements in the order presented above. QUESTION 1: The empirical formula for this compound is . QUESTION 2: The molar mass for this compound is 117.2 g/mol. The molecular formula for this compound is .

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, baileysosmart
The diagram shows the positions of the sun, moon and earth during spring tides, when the high tides are at their highest and low tides at their lowest. what is it about these positions that causes these high and low tides?
Answers: 3

Chemistry, 22.06.2019 02:10, fvmousdiana
Determine the percent sulfuric acid by mass of a 1.61 m aqueous solution of h2so4. %
Answers: 2

Do you know the correct answer?
A compound is found to contain 9.224 % boron and 90.74 % chlorine by mass. To answer the question, e...
Questions in other subjects:

Mathematics, 29.01.2020 00:48



History, 29.01.2020 00:48



Computers and Technology, 29.01.2020 00:48

History, 29.01.2020 00:48

History, 29.01.2020 00:48

Social Studies, 29.01.2020 00:48


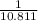 =0.85 mol (Mass value of boron=10.811)
=0.85 mol (Mass value of boron=10.811)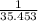 =2.559 mol.(Mass value of chlorine=35.453)
=2.559 mol.(Mass value of chlorine=35.453)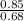 =1
=1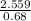 =3
=3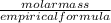
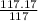 ≈1
≈1



