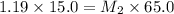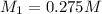Chemistry, 26.03.2020 21:08, mangowammy
A student prepared a stock solution by dissolving 10.0 g of KOH in enough water to make 150. mL of solution. She then took 15.0 mL of the stock solution and diluted it with enough water to make water to make 65.0 mL of a final solution. What is the concentration of KOH for the final solution

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 23.06.2019 02:00, sakria2002
What is the difference between a substance "getting wet" and "being dissolved" in a liquid at the particulate level?
Answers: 3
Do you know the correct answer?
A student prepared a stock solution by dissolving 10.0 g of KOH in enough water to make 150. mL of s...
Questions in other subjects:


Chemistry, 02.10.2020 14:01


Mathematics, 02.10.2020 14:01

English, 02.10.2020 14:01


Mathematics, 02.10.2020 14:01

Mathematics, 02.10.2020 14:01



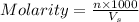
 = volume of solution in ml = 150 ml
= volume of solution in ml = 150 ml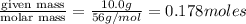
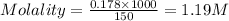
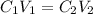
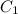 = molarity of stock solution = 1.19 M
= molarity of stock solution = 1.19 M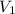 = volume of stock solution = 15.0 ml
= volume of stock solution = 15.0 ml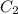 = molarity of diluted solution = ?
= molarity of diluted solution = ?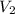 = volume of diluted solution = 65.0 ml
= volume of diluted solution = 65.0 ml