
Chemistry, 26.03.2020 21:12, torquishag
Consider the following reaction in which all reactants and products are gases. 1.00 mol of A and 2.00 mol of B are placed in a 5.0-liter container. After equilibrium has been established, 0.50 mol of D is present in the container. Calculate the equilibrium constant, Kc, for the reaction.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, Angelanova69134
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2


Chemistry, 22.06.2019 07:30, isalih7256
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1

Chemistry, 22.06.2019 18:30, lattimorekeonna1
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
Do you know the correct answer?
Consider the following reaction in which all reactants and products are gases. 1.00 mol of A and 2.0...
Questions in other subjects:

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00





Chemistry, 11.02.2021 01:00

Physics, 11.02.2021 01:00


![K_{c} = \frac{[D]}{[A][B]}](/tpl/images/0565/8107/95178.png) =
= 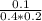 =1.25
=1.25



