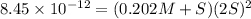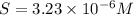Chemistry, 26.03.2020 02:53, erbnichole
Consider the dissolution of AB(s): AB(s)⇌A+(aq)+B−(aq) Le Châtelier's principle tells us that an increase in either [A+] or [B−] will shift this equilibrium to the left, reducing the solubility of AB. In other words, AB is more soluble in pure water than in a solution that already contains A+ or B− ions. This is an example of the common-ion effect. The generic metal hydroxide M(OH)2 has Ksp = 8.45×10−12. (NOTE: In this particular problem, because of the magnitude of the Ksp and the stoichiometry of the compound, the contribution of OH− from water can be ignored. However, this may not always be the case.) Part A What is the solubility of M(OH)2 in pure water? Express your answer with the appropriate units. View Available Hint(s) nothing nothing Part B What is the solubility of M(OH)2 in a 0.202 M solution of M(NO3)2? Express your answer with the appropriate units. View Available Hint(s) nothing nothing

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, blondieb1722
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2

Chemistry, 22.06.2019 18:00, faithabossard
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 23.06.2019 00:00, alisonsolis155
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3
Do you know the correct answer?
Consider the dissolution of AB(s): AB(s)⇌A+(aq)+B−(aq) Le Châtelier's principle tells us that an inc...
Questions in other subjects:


English, 20.09.2020 17:01

English, 20.09.2020 17:01

History, 20.09.2020 17:01

Mathematics, 20.09.2020 17:01





English, 20.09.2020 17:01


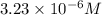 is the solubility of
is the solubility of 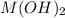 in a 0.202 M solution of
in a 0.202 M solution of 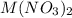 .
.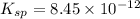
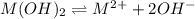
![K_{sp}=[M^{2+}][OH^-]^2](/tpl/images/0564/5689/a461b.png)
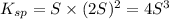
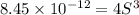
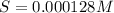
![[M^{2+}]=0.202 M+S](/tpl/images/0564/5689/48853.png)