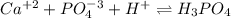Chemistry, 26.03.2020 01:10, yogibear9998
Identify the effect of increasing acidity on the solubility of the given compounds. ca3(po4)2

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 23.06.2019 02:30, kieante01
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1
Do you know the correct answer?
Identify the effect of increasing acidity on the solubility of the given compounds. ca3(po4)2...
Questions in other subjects:

Computers and Technology, 14.11.2019 21:31


History, 14.11.2019 21:31


Mathematics, 14.11.2019 21:31



Mathematics, 14.11.2019 21:31



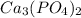 will increase.
will increase.