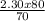Chemistry, 25.03.2020 06:56, Nolife012603
You select a 150.0 ml volumetric flask to which you add 80.00 mL of a 2.30 M solution of the weak acid you just selected. To finish preparing your buffer you must now add the sodium salt of the conjugate base. How many grams of the sodium salt of your conjugate base must you add so that when you finally fill the flask to the mark with deionized water the buffer will have a pH of 8.50.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:00, juliannxkim
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 09:00, oliviacolaizzi
Ineed to find the answer of this question because i dont understand it
Answers: 1

Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
Do you know the correct answer?
You select a 150.0 ml volumetric flask to which you add 80.00 mL of a 2.30 M solution of the weak ac...
Questions in other subjects:




Social Studies, 07.12.2020 22:30


Chemistry, 07.12.2020 22:30



Chemistry, 07.12.2020 22:30

Mathematics, 07.12.2020 22:30