

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 20:00, 20calzoy
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 22.06.2019 20:30, lexibyrd120
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2
Do you know the correct answer?
The heat change that accompanies the formation of 1.00 mol of carbon dioxide from its elements is −3...
Questions in other subjects:

Mathematics, 25.03.2021 02:10


Social Studies, 25.03.2021 02:10

Mathematics, 25.03.2021 02:10

Mathematics, 25.03.2021 02:10


English, 25.03.2021 02:10


Geography, 25.03.2021 02:10


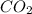 is -373.7 KJ/mol. Let us consider the required Heat Exchange at 0.631 mol of
is -373.7 KJ/mol. Let us consider the required Heat Exchange at 0.631 mol of 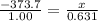
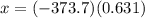
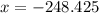 KJ/mol
KJ/mol




