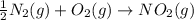Chemistry, 25.03.2020 03:38, lexieprochaskaaa
Write an equation for the formation of one mole of NO2(g) from its elements in their standard states. Express your answer as a chemical equation using fractional coefficients as needed. Identify all of the phases in your answer.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:10, GreatBaconGamer
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1

Chemistry, 22.06.2019 22:30, kiera2599
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1


Chemistry, 22.06.2019 23:30, sanociahnoel
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
Do you know the correct answer?
Write an equation for the formation of one mole of NO2(g) from its elements in their standard states...
Questions in other subjects:

Mathematics, 22.11.2019 06:31

English, 22.11.2019 06:31