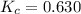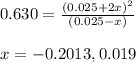Chemistry, 25.03.2020 02:08, gunnatvinson
The value of kc for the following reaction is 0.630 at 409 K N2O4(g) --> 2NO2(g) if a reaction vessel at that temperature intitially contains 0.0250 M NO2 and 0.0250 M N2O4, what is the concentration of NO2 at equilibrium

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, lpssprinklezlps
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

Chemistry, 22.06.2019 12:10, kaitlynbernatz2778
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3
Do you know the correct answer?
The value of kc for the following reaction is 0.630 at 409 K N2O4(g) --> 2NO2(g) if a reaction ve...
Questions in other subjects:

Mathematics, 04.07.2019 14:30



History, 04.07.2019 14:30

History, 04.07.2019 14:30




Mathematics, 04.07.2019 14:30

Mathematics, 04.07.2019 14:30


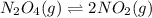
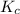 for above equation follows:
for above equation follows:![K_c=\frac{[NO_2]^2}{[N_2O_4]}](/tpl/images/0562/2368/271f5.png)