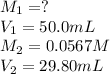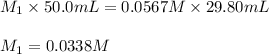Chemistry, 25.03.2020 00:28, itzdulceee
Acetic acid (CH3CO2H) is the principal component in the vinegar. What is the molarity of an acetic acid solution if a titration of 50.00 mL of the acetic acid solution requires 29.80 mL of 0.0567 M NaOH to reach the equivalence point?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, homeschool0123
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1



Chemistry, 22.06.2019 20:00, aksambo4707
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
Do you know the correct answer?
Acetic acid (CH3CO2H) is the principal component in the vinegar. What is the molarity of an acetic a...
Questions in other subjects:



English, 27.09.2021 16:10

French, 27.09.2021 16:10


English, 27.09.2021 16:10

Biology, 27.09.2021 16:10

English, 27.09.2021 16:10

Mathematics, 27.09.2021 16:10


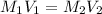
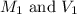 are the molarity and volume of acetic acid.
are the molarity and volume of acetic acid.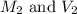 are the molarity and volume of NaOH.
are the molarity and volume of NaOH.